The SARS coronavirus came out of nowhere. It is an example of the speed with which a zoonotic disease can leap to the fore of international attention. We were completely surprised. Fortunately, the virus was not quite as virulent as first feared, and it burned itself out before causing greater havoc. This is an important misconception about the SARS episode. Yes, the virus was identified and sequenced in a hurry, but all our technology was of little help in responding. We got lucky.
According to an epidemiological modeling paper published a year after the epidemic:
We conclude that the control of SARS through the use of simple public health measures was achieved because of the efficacy with which those measures were introduced and the moderate transmissibility [R0] of the pathogen coupled with its low infectiousness prior to clinical symptoms[theta].
[Fraser, et al., PNAS | April 20, 2004 | vol. 101 | no. 16 | 6146-6151]
That is, it is quite possible that the much vaunted public health measures used to battle the SARS virus were only effective because the virus wasn't actually that bad. Not to minimize the death, disease, and the at least US$ 50 billion in economic damage, but it could well have been a lot worse.
This conclusion comes out of a modeling paper, and describes the development of a methodology similar to the one now being used to plan responses to a pandemic flu outbreak. Unfortunately, there isn't a great deal of data to constrain the model, and as Tara O'Toole and her colleagues at the Center for Biosecurity at UPMC point out (PDF), present monitoring and quarantine policy is simply "inconsistent with available scientific understanding of the nature of person-to-person disease transmission." There is also a distinct question as to how far we should trust the model. Fortunately, we can look at distinct historical events to figure out whether preparations are on the right track.
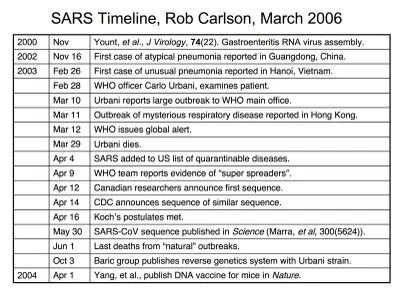
Below is a time line of events starting in the fall of 2002, when the SARS coronavirus first emerged. (Sources: WebMD, Science, Nature, PNAS, Journal of Virology.)
Note that while the virus appears to have emerged in November of 2002, it wasn't until February of 2003 that Carlo Urbani saw his first patient. The diagnosis in November is entirely forensic in nature. That is, working backwards this seems to be when the virus first jumped to humans. Koch's postulates, which must be met in order to conclusively link a pathogen and the disease it causes, are not met until the middle of April. The sequence is then announced at the height of the pandemic, though it isn't published until after most deaths from the initial outbreak have already happened. Ralph Baric's group at UNC publishes the first paper in October demonstrating control of the virus in the lab, which is a prerequisite to doing any biology, figuring out how the virus works, and developing vaccines. (Ralph told me at a meeting last week they were ready go to with the paper a few months before it came out. This delay was probably due to academic publishing BS, as far as I can tell, though it might have gone faster if people were still dying at that point.) The first vaccine takes another year to test and publish (some of this interval is also due to the dynamics of academic publishing, but the point remains).
Thus the pandemic was basically over by the time we could do any biology and even start to think about vaccines. But Ralph Baric wouldn't have been able to move as fast as he did in 2003 had he not worked out the packaging strains for the coronavirus reverse genetics system years earlier, published in November of 2000 (Yount, et al., J. Virology). As it happened, Ralph thought the viruses were interesting, and he put in the time and effort to sort out how to work with them in the lab. It is only through Ralph's efforts that the rest of us have the good fortune to know as much as we do now about the virus.
What about the next time? The reverse genetics system for influenza was published in 1999, and we are still trying to figure out how the virus works. If a flu pandemic hits we might, just maybe, be ready with useful vaccines and antivirals, particularly if the FDA's new flu vaccine licensing rules turn out to be as well thought out as has been reported. Future flu pandemics are a near certainty, if only because we have historical examples. Thus there is a clear motivation to get ready for the next one. We have a choice about whether to prepare, and the flu is an understood threat. But what about the next true surprise, the next SARS coronavirus? And what if it is just a little bit worse than SARS?
We have to be able to detect the threat, understand it, and respond much more rapidly than is now possible.